Dermapose Refresh Stand
a reusable, autoclave-safe holder that secures the Refresh device during fat processing.
Corporate Headquarters:
Bimini Health Tech
8400 Belleview Drive, Suite 125
Plano, TX 75024
+1 (858) 348-8050
Research & Development:
Bimini Health Tech
420 Stevens Ave, Suite 220
Solana Beach, CA 92075
+1 (858) 348-8050
Dermapose® allows you to harvest, purify, resize, and inject microfat with a single closed system, empowering you with a practical, office-based procedure.
a single-use all-in-one fat harvesting, purifying, and microsizing device.
Learn More About Dermapose Refresh™
a reusable, autoclave-safe holder that secures the Refresh device during fat processing.
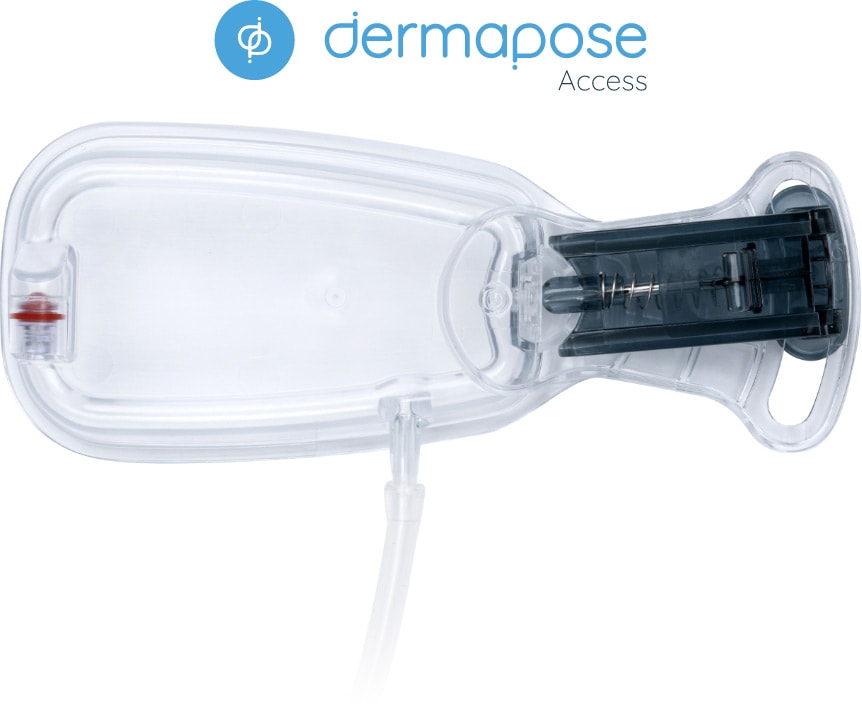
an optional single-use guided fat harvesting access system that integrates with Refresh.
Learn More About Dermapose Access™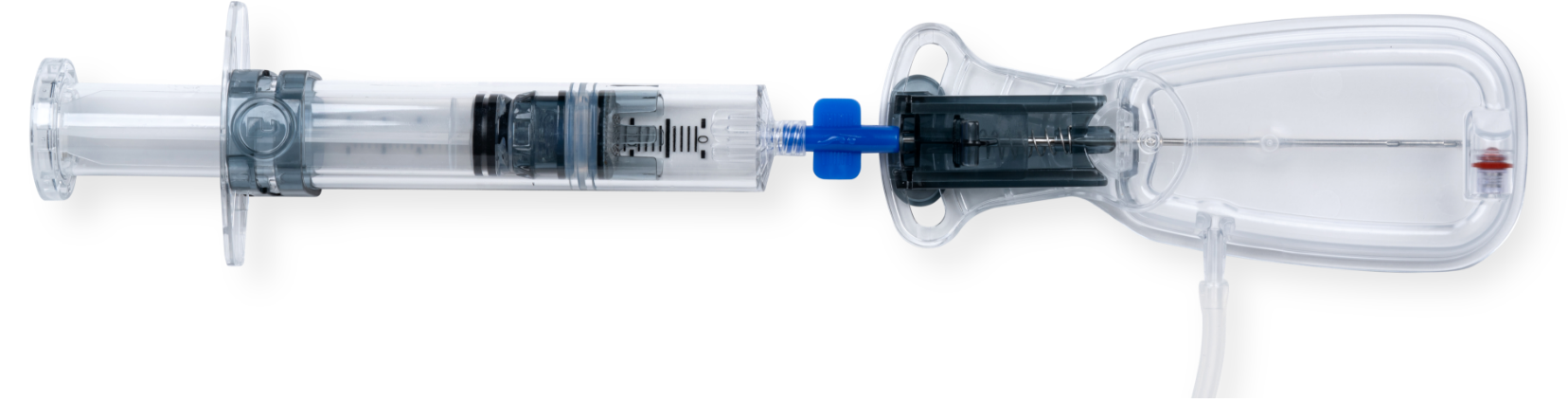
This sterile, single-use, dual-chamber device gives you the power to produce approximately 15 cc of pure, viable microfat ready for reinjection.
GET DERMAPOSE refresh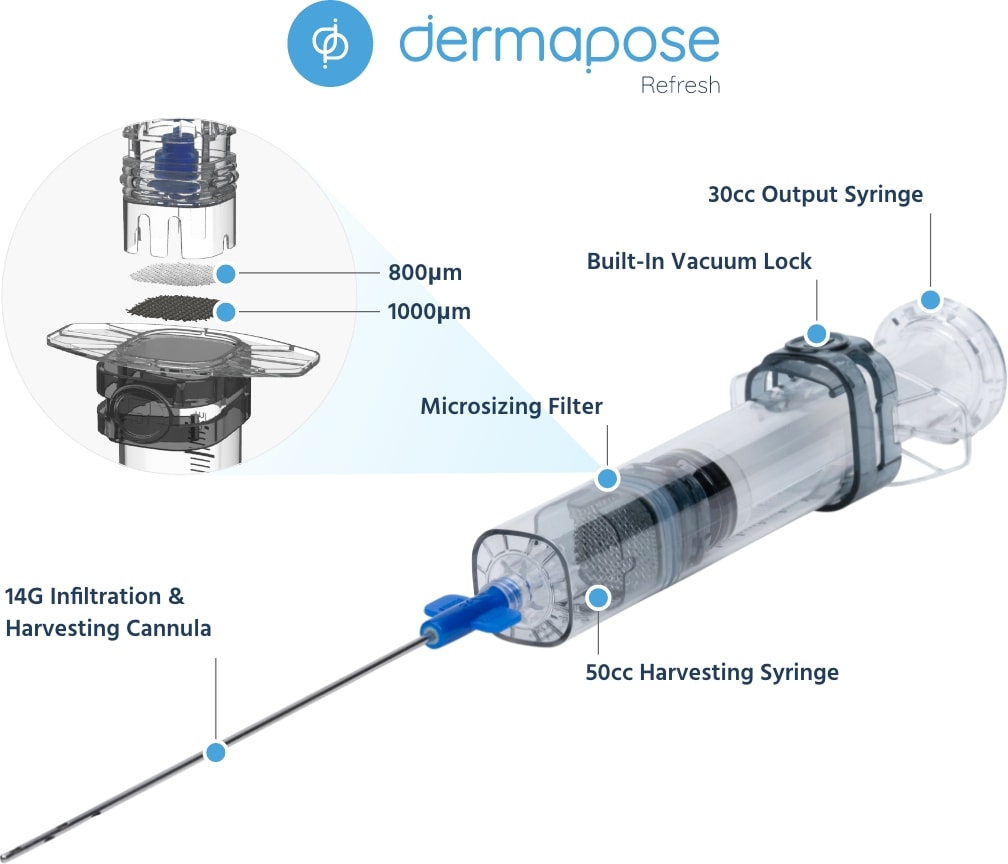
Dermapose Refresh makes microfat grafting quick and easy in an office setting. Built-in 800 and 1000-micron filters uniformly resize adipose tissue so that it’s small enough to fit through 18 to 21G injectors without harming adipocytes and regenerative cells.
This innovative, dual-chambered syringe system makes it easy to produce high-quality, small-volume microfat grafts in less than an hour.
All-in-one device for harvesting, purifying, resizing, and transferring microfat that is consistent and reproducible.
Fat remains in a single-use, dual-chambered, closed system from harvest to transfer.
Obtain up to 30 cc of pure microfat in under 60 minutes.
The optional Dermapose Access device simplifies what can be a challenging step for an in-office fat transfer process: harvesting fat. This sterile, single-use, closed system provides easy access to subcutaneous fat for harvesting with Dermapose Refresh. Access improves small-volume fat harvesting by:
In aesthetic medicine, microfat is a superior injectable that is small enough to flow through 18 to 21-gauge injectors. It adds structural support under the skin and may improve skin quality with innate regenerative cells and growth factors. Advantages over synthetic fillers include:
“Being able to harvest, purify and microsize 15mL of adipose tissue within a sterile closed-system syringe opens fat grafting up to patients in an office setting. Dermapose Refresh is the ideal way to obtain microfat in an easy, reproducible manner.”
E. Daniels, M.D.
Dermapose makes small-volume fat transfer a superior alternative to synthetic dermal fillers for providing a variety of cosmetic benefits, including:
“I have used a wide variety of fat processing and decant methods over the years and I always use Puregraft for my breast cases because it’s (a) FAR more predictable intake and reduction in fat necrosis. I have been using it for 8 years.”
Paul Ruff, M.D. FACS, Washington, DC
Smooth away wrinkles and nasolabial folds; fill in sunken cheeks, hollows around the eyes, and withered hands.
Plump up and add definition to thin, deflated lips.
Elevate depressions, smooth out unevenness, and soften damaged tissue.
May help improve skin firmness and texture on the face, eye area, lips, neck, and hands.
PureGraft is a state-of-the-art fat filtering system, available in 50-, 250-, and 850-mL sizes, that delivers 97%1 pure fat every time.
EXPERIENCE PUREGRAFT
Bimini Health Tech empowers you to help patients tap their true potential. Developed through scientific rigor and superior processes, our Dermapose products make office-based microfat grafting simple, effective, and predictable, leading to consistently superior results. Bimini continues to honor its commitment to delivering world-class technology you can trust.
Dermapose Refresh and Dermapose Access Are Suction Lipoplasty Systems
Dermapose Refresh is a sterile medical device intended for the closed-loop processing of lipoaspirate tissue in medical procedures involving the harvesting, concentrating, and transferring of autologous adipose tissue harvested with a legally marketed lipoplasty system. The device is intended for use in the following surgical specialties when the transfer of harvested adipose tissue is desired: orthopedic surgery, arthroscopic surgery, neurosurgery, gastrointestinal and affiliated organ surgery, urological surgery, general surgery, gynecological surgery, thoracic surgery, laparoscopic surgery, and plastic and reconstructive surgery when aesthetic body contouring is desired. Only legally marketed accessory items, such as syringes, should be used with the system. If harvested fat is to be transferred, the harvested fat is only to be used without any additional manipulation. See the Instructions for Use that accompanies the product for important Warnings, Precautions, and Directions.
The Dermapose Access fat harvest system is intended for use in aesthetic body contouring. See the Instructions for Use that accompanies the product for important Warnings, Precautions, and Directions.
1. 500-003-01, REV A. White Paper. Comparison of Three Fat Graft Preparation Methods: Gravity Separation, Centrifugation, and the Puregraft® System.